
US-listed shares in Alterity Therapeutics (NASDAQ:ATHE) have soared post and premarket after the company reported positive data from animal testing of its leading drug candidate for Multiple System Atrophy…
The company stated that the drug, ATH434, has been selected for presentation at the 2020 International Congress of Parkinson’s Disease and Movement Disorders, as well as the American Neurological Association’s 2020 Annual Meeting.
“New data independently confirms and extends laboratory findings and expands safety profile of ATH434,” said Alterity in its statement.
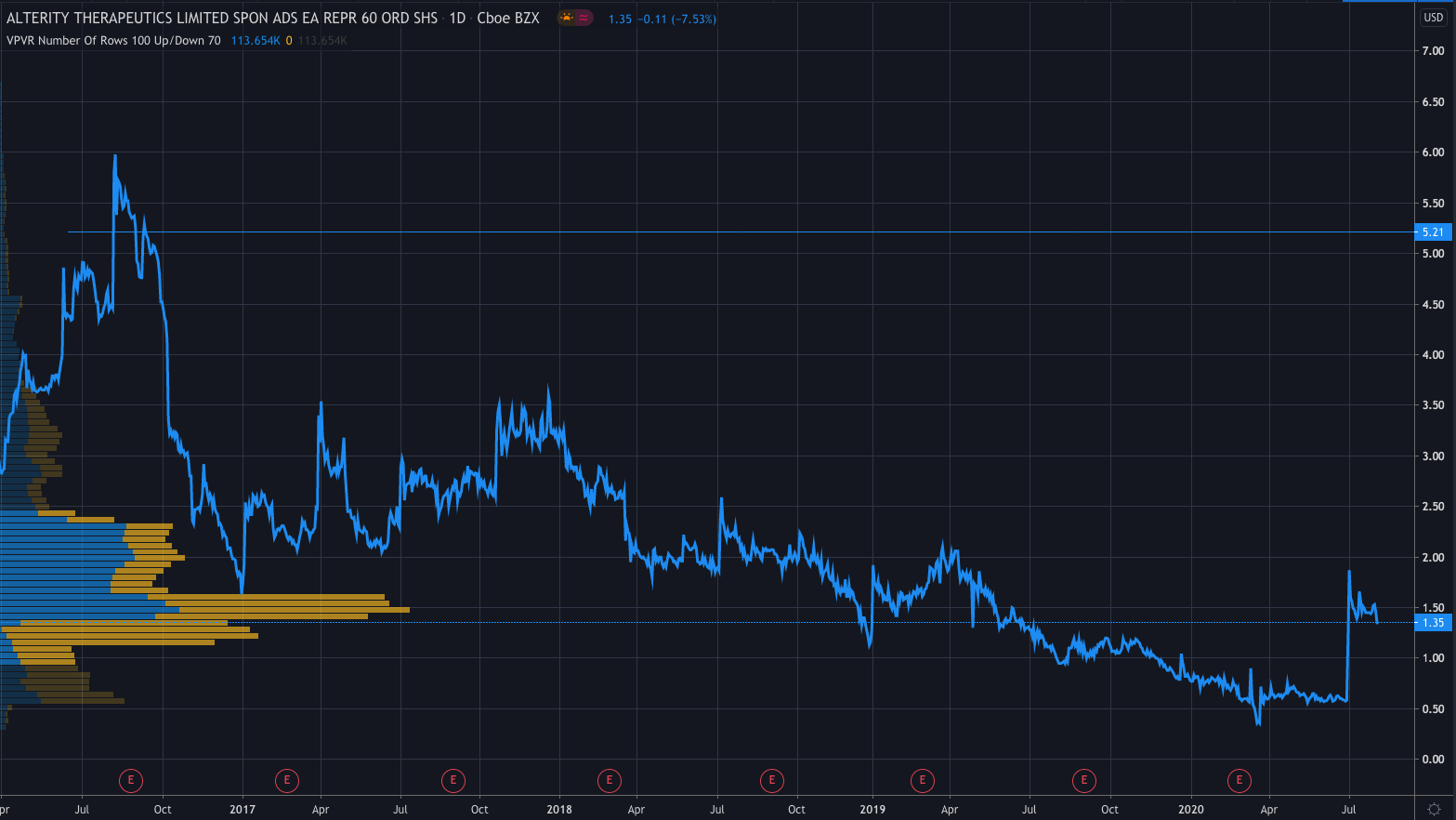
After the announcement, shares in the company skyrocketed and are currently trading at $5.21 premarket, after a 285.93% move higher, taking it to levels not seen since September 2016.
The company’s share price has fallen significantly in the last five years and its no surprise given its lack of profitability.
“Drug-induced QT prolongation can pose a significant risk for patients, so a clean bill of health on this safety measure is important as we advance to Phase 2. In searching for new treatments to modify disease progression, we need to develop agents that are as safe as possible,” said Alterity’s Chief Medical Officer, Dr David Stamler.
Alterity will also present cardiac safety data from its phase one study of ATH424, which will be the first time this type of information is shared amongst international physicians and researchers in neurological disorders.
- Trade Stocks With IG Markets
