Shares of Moderna Inc (NASDAQ: MRNA) were up premarket after the UK approved its COVID-19 vaccine for use in the country that imposed a lockdown earlier this week in efforts to control the rising coronavirus cases.
The biotech company said that it appreciates the confidence shown by the UK MHRA in its COVID-19 vaccine, which is an important step in the global fight against the virus that had become a global pandemic.
Moderna’s vaccine is composed of two doses administered 28 days apart and is approved for people aged 18 year and older. The vaccine has an efficacy rate of 94% and could help reverse the growing coronavirus cases in the UK.
 WELCOME BONUS - Free Share Bundle When You Invest £50!
Open a UK Investment Account: Shares, ISAs, Managed Portfolio
Invest in 15,000+ shares and ETFs. Open an account now, invest at least £50, and you’ll get a free share bundle worth between £40 and £200. T&Cs apply.
IG
View Offers
WELCOME BONUS - Free Share Bundle When You Invest £50!
Open a UK Investment Account: Shares, ISAs, Managed Portfolio
Invest in 15,000+ shares and ETFs. Open an account now, invest at least £50, and you’ll get a free share bundle worth between £40 and £200. T&Cs apply.
IG
View Offers
 Empfohlener Broker
Multi Asset Platform
Empfohlener Broker
Multi Asset Platform
The UK first approved the Pfizer/BionTech vaccine on December two followed by Oxford University/AstraZeneca vaccine on December 30, making Moderna’s vaccine the last to receive approval in the country.
Moderna’s vaccine has so far received approval from the European Union, the US, Israel and Canada, but its stock price has been falling ever since early December 2020 despite the recent approvals.
It is not yet clear whether Moderna’s vaccine is effective against the two virulent strains of the COVID-19 vaccine that were discovered in South Africa and the UK.
Pfizer and BionTech today said that in vitro studies have shown that its vaccine effectively neutralizes the mutated variants of the SARS-CoV-2 virus discovered in the two countries above.
Moderna share price
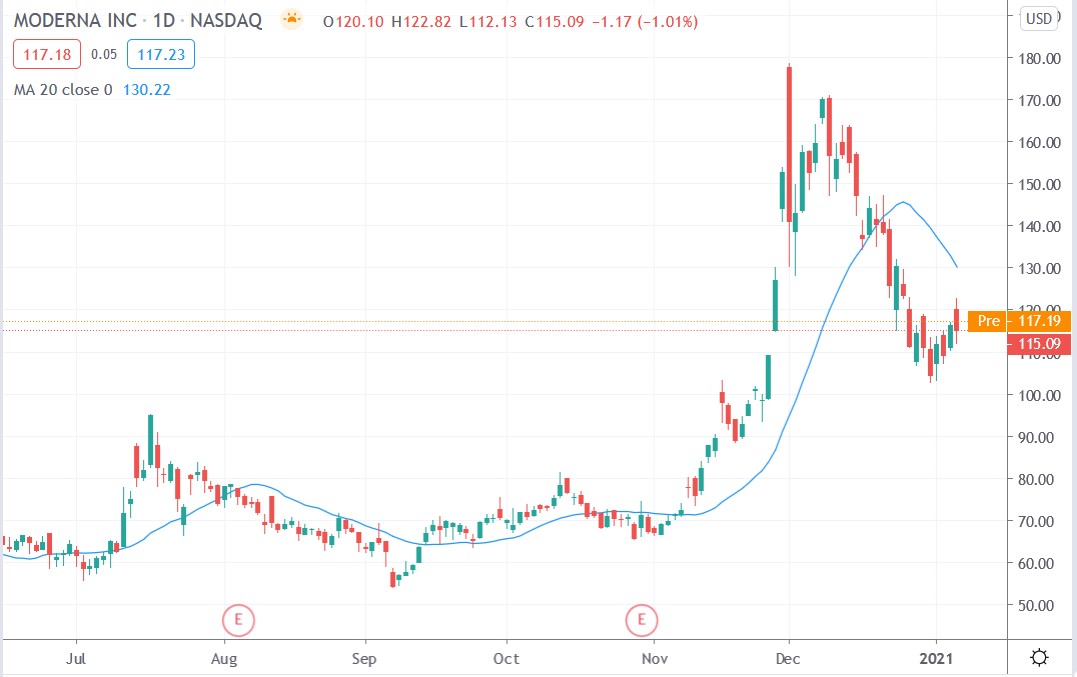
Moderna shares were up 1.82% premarket to trade at $117.23 having slightly higher from Thursday’s closing price of $115.09.
