Shares of Synairgen Plc (LON: SNG) today rallied 15.3% after the company released positive phase II clinical trial results for SNG001, a wholly-owned inhaled formulation of interferon beta for COVID-19 patients, on older populations with significant co-morbidity (COPD).
The interim analysis of its exploratory Phase II clinical trial of inhaled SNG001 in Chronic Obstructive Pulmonary Disease (COPD) patients with a confirmed respiratory viral infection showed that it was well tolerated in older populations with a mean age of 66 years.
Synairgen had announced positive phase I clinical trial results for SNG001 on 20th July.
 WELCOME BONUS
Trade Thousands Of Global Markets
Demo account, mobile app and multi-platform access
IG
Visit Site
WELCOME BONUS
Trade Thousands Of Global Markets
Demo account, mobile app and multi-platform access
IG
Visit Site
 Empfohlener Broker
Multi Asset Platform
Empfohlener Broker
Multi Asset Platform
Richard Marsden, CEO Synairgen, said: “COPD exacerbations are the second most common cause of unplanned hospitalisation in England, behind cardiovascular disease…Overall, SNG001 has now been shown to raise the body’s natural viral defences when challenged by a wide variety of respiratory viruses, indicating that it could be an important treatment in the coming virus season, where there may be coinfection with influenza and other viruses alongside COVID-19.
Synairgen share price
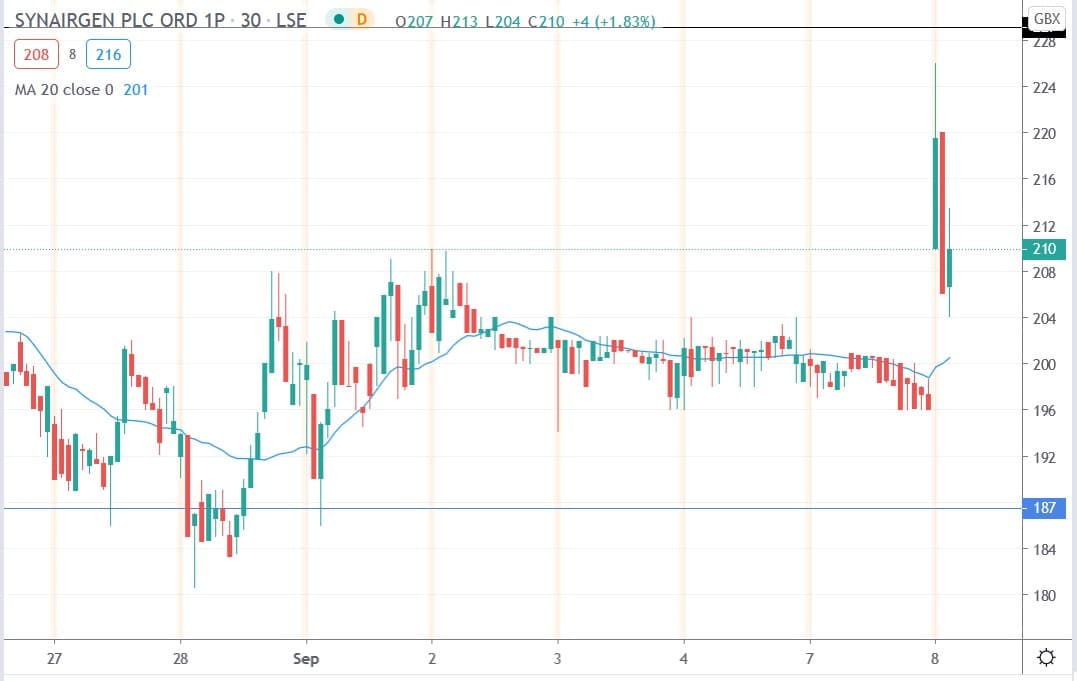
Synairgen shares today surged 15.3% to trade at 226p having closed yesterday’s session trading at 196p.
People who read this also read:
- Here are our latest trending stories
- Trade shares with the highly-rated IG UK
- Here are the 7 best shares to buy in 2020
